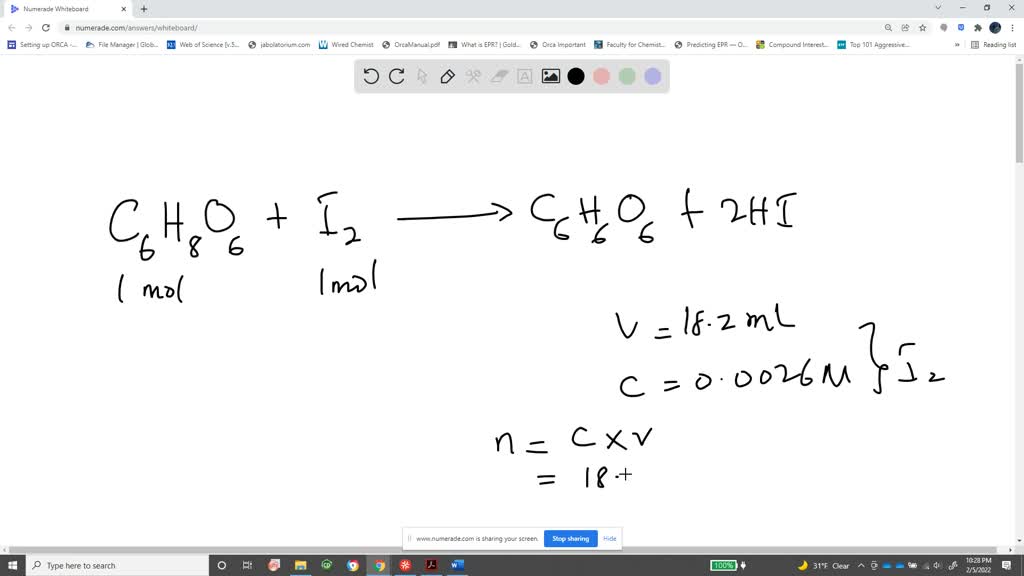
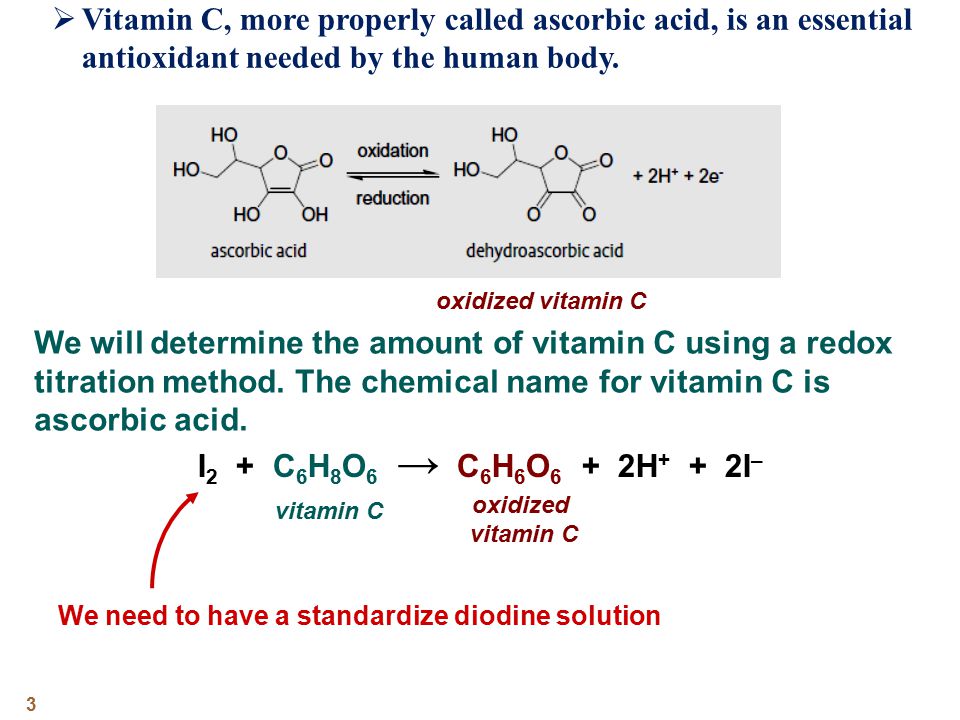
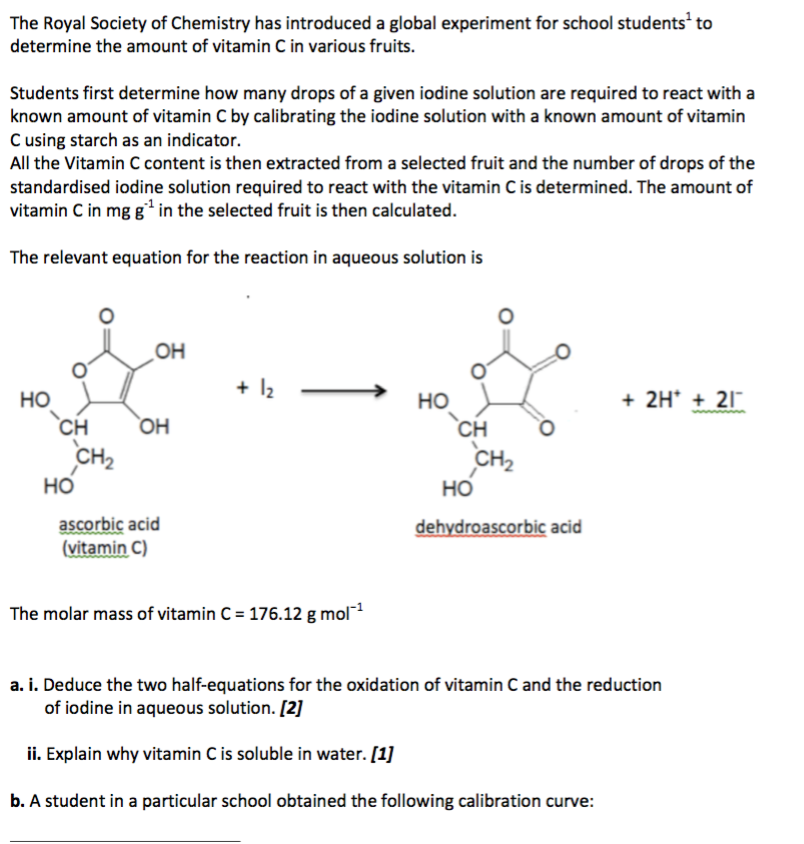
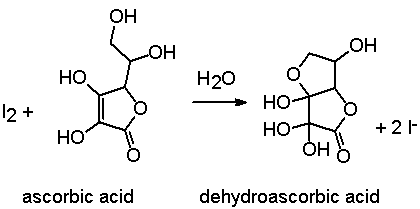
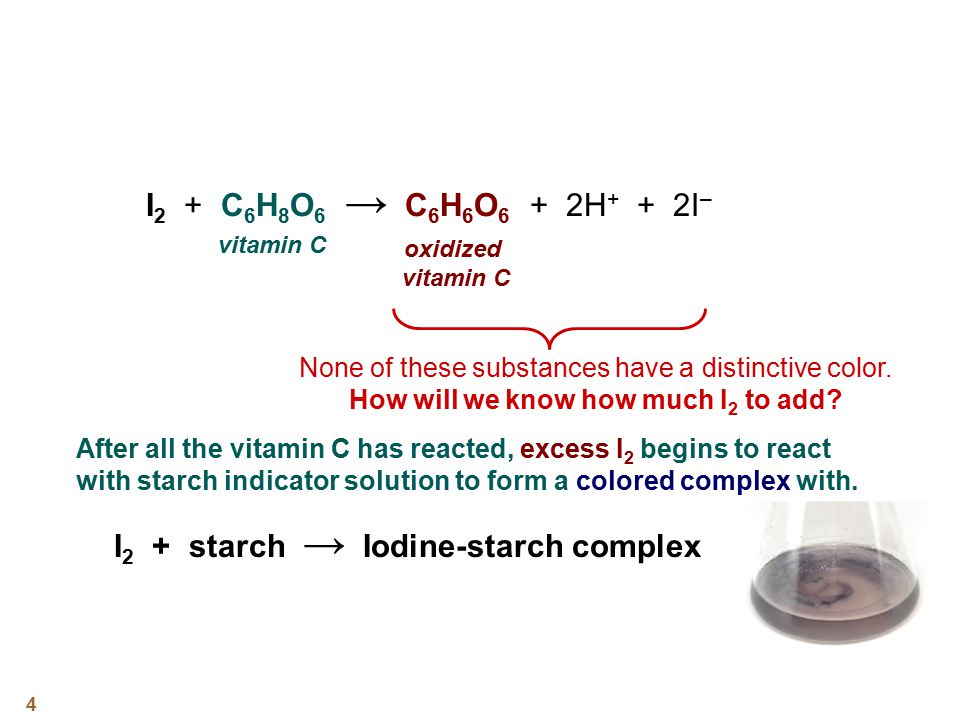
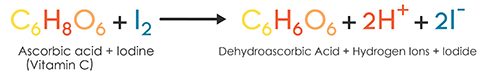
SOLVED:The amount of ascorbic acid (vitamin C) in fruit juice is determined by a titration using a redox reaction. Iodine is the titrant, but because iodine solutions in water are unstable, the

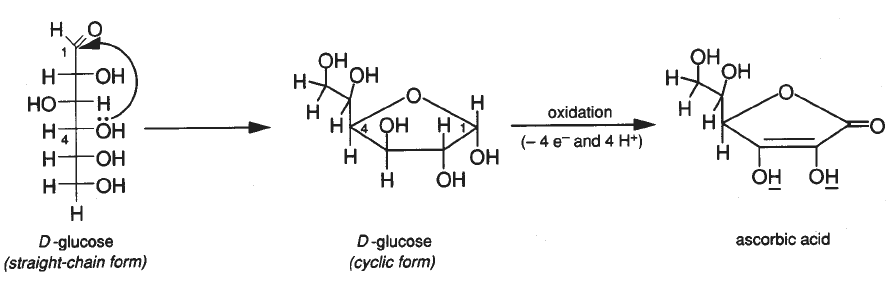
Ascorbic acid (vitamin C), along with having many other reputed properties, acts as an antioxidant. The following equation illustrates its antioxidant properties. H_2C_6H_6O_6 \to C_6H_6O_6 + H_2(g) What is an antioxidant? Assign

OneClass: 1. Ascorbic acid (vitamin C) in a juice drink can be determined by means of a constant-curr...

2.8: Kinetics- Iodine Clock using Vitamin C and the Calculation of Initial Rate-Home - Chemistry LibreTexts

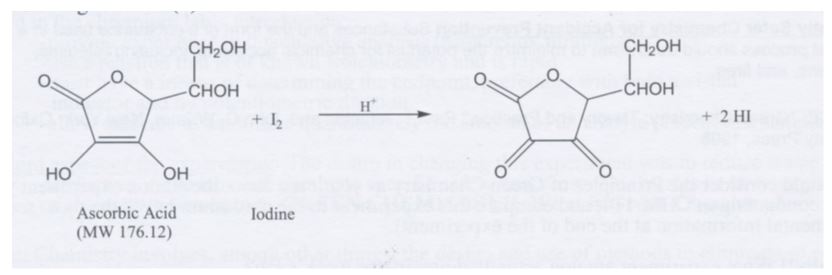
SOLVED: Pre-lab Balance the reaction of ascorbic acid with iodine. Use the method of your choice: mark) CoHsOs(aq) [z(aq) CoHsOs(aq) (aq) (acidic) The recommended daily allowance of' vitamin € is 60 mg