REG-97 Požadavky na předkládání kvalitních národních překladů informací o přípravku a na používanou terminologii

Missing data, clade support and “reticulation”: the molecular systematics of Heliconius and related genera (Lepidoptera: Nymphalidae) re‐examined - Brower - 2018 - Cladistics - Wiley Online Library

Utility of B-Factors in Protein Science: Interpreting Rigidity, Flexibility, and Internal Motion and Engineering Thermostability | Chemical Reviews

Rice SUB1A constrains remodelling of the transcriptome and metabolome during submergence to facilitate post‐submergence recovery - Locke - 2018 - Plant, Cell & Environment - Wiley Online Library

Genotoxic Impurities in Pharmaceutical Manufacturing: Sources, Regulations, and Mitigation | Chemical Reviews

PDF) Thermal adaptation in the honeybee ( Apis mellifera ) via changes to the structure of malate dehydrogenase

Comparing mutagenesis and simulations as tools for identifying functionally important sequence changes for protein thermal adaptation | PNAS
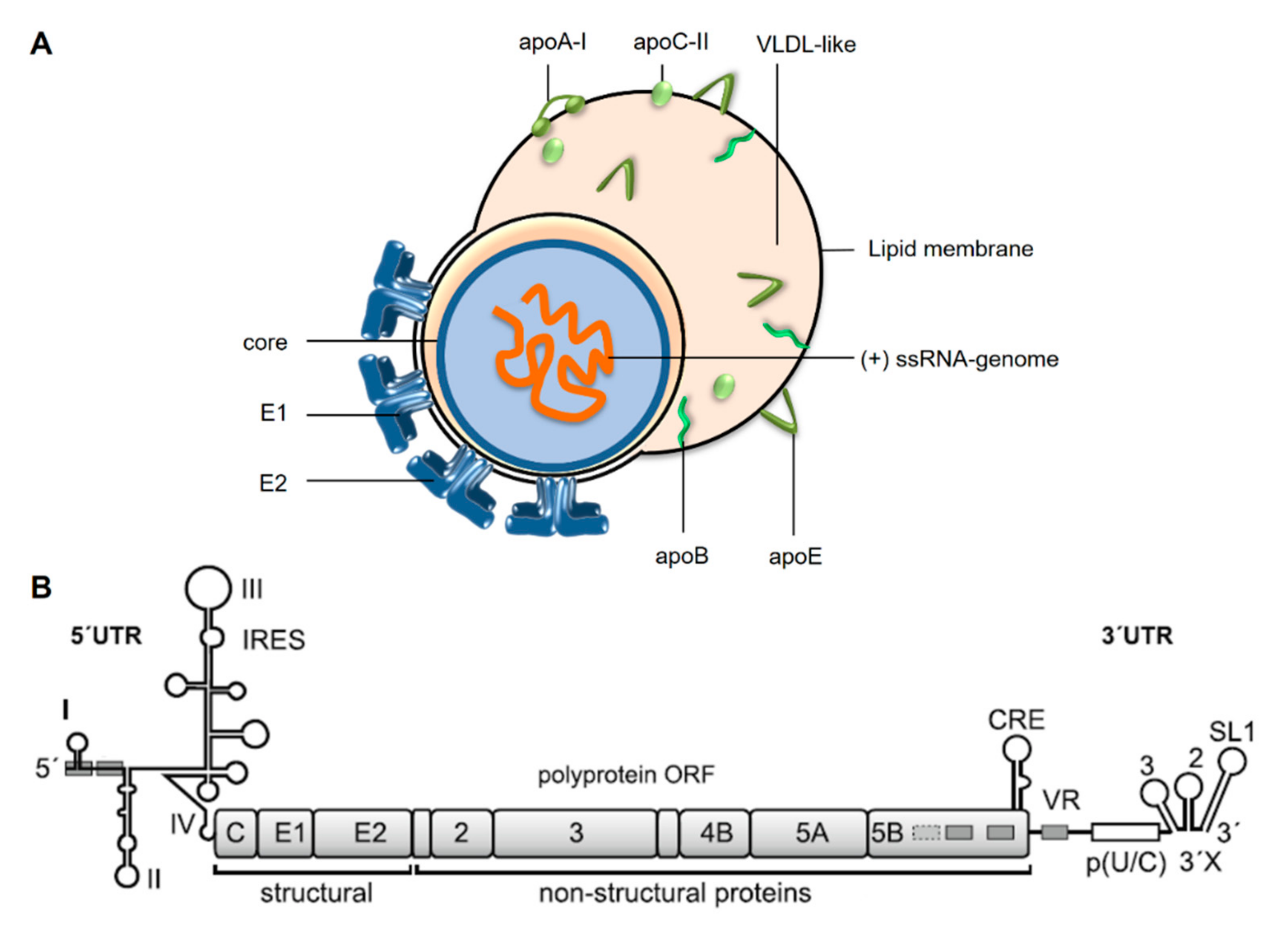
Cells | Free Full-Text | Hepatitis C Virus Downregulates Core Subunits of Oxidative Phosphorylation, Reminiscent of the Warburg Effect in Cancer Cells



